
Slides supporting 'The Perth Group revisits the existence of HIV' paper

This is the electron micrograph published by Montagnier.
In this slide the broad, uninterrupted structure in the bottom half, is a portion of an umbilical cord lymphocyte. On its surface are some blebs (buds) and nearby are a few, cell free particles. These appearances were reported as “virus-producing cord lymphocytes [showing] various stages of particle budding at the cell surface”.
Montagnier classified these particles as genus type C particles of the subfamily Oncovirus. In fact in the abstract of the paper these are called “a typical type-C tumor virus” (=Oncovirus). Later it became known that Gallo wrote the abstract of the Montagnier paper but also, in his own 1984 isolation papers, Gallo also classified the “new retrovirus” within the same genus, type-C particles.

Here it is a retroviral particle, a type-C particle, which is found in the majority of placentas. On the left is the low power magnification and on the right the high powered magnification.
Panem S: C Type Virus Expression in the Placenta. Curr Top Pathol 1979, 66:175-189

Here is an image of a placental type C particle alongside the Montagnier’s 1983 HIV particles. Those reported in 1983 as “typical type-C” particles.
Who can say whether Montagnier’s cultures would have contained or produced such particles even if they had been made without lymphocytes from BRU? Without controls it is impossible to know.
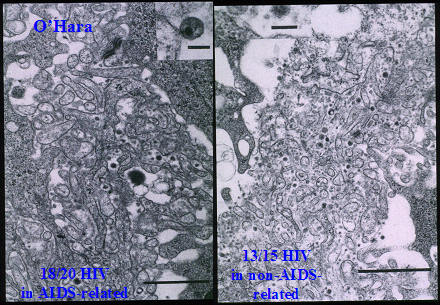
This research showed that "HIV particles" were present in 18/20 (90%) of patients with enlarged lymph nodes attributed to AIDS. However, identical particles were found in 13/15 (87%) of patients with enlarged lymph nodes not attributed to AIDS and at no risk for developing AIDS.

This slide was published in 1961 and confirms it is possible to take an electron micrograph of the 1.16 gm/ml band and show purified, retroviral particles. These are particles of the Rous sarcoma virus, an animal retrovirus discovered by Rous in 1911. Notice that unlike many images of HIV, this EM has a size bar measuring 1000 nanometres.
For over twenty years we have been asking for similar evidence in regard to HIV. That is, evidence that what Montagnier and Gallo called “purified” virus, consists of retroviral particles and nothing but retroviral particles.
Nobody responded but in 1997, two groups of researchers, one a Franco/ German collaboration and another from the US National Cancer Institutes, each published a paper in the March issue of the journal Virology.

So here is the first electron micrograph, from the Franco/German group*, to show what is actually in this material.
The upper part labeled (a) is the 1.16 gm/ml band from infected cells, that is H9 cells infected with HIV. This middle part, labeled (b), is again the 1.16 gm/ml band, again from infected cells, but this time cells were obtained from normal individuals.
The bottom part represents the 1.16 gm/ml band obtained from non-infected cell cultures.
You do not need to be a scientist or HIV expert to realise that, whatever these pictures represent, it is not purified anything. In something that is purified every object looks the same as every other object. Just think of a box of apples. In fact the authors of this paper labeled the first section (a) “Purified vesicles from infected H9 cells”, and the second section (b) as “activated peripheral blood mononuclear cells”. Although this paper is about HIV preparations they did not label their pictures “purified HIV” because they could not. In other words, after fourteen years HIV experts themselves confirm there is no such thing as purified HIV.
If we examine the pictures in more detail what do we see? Most of the material is cellular fragments and in some places the authors have placed arrows to indicate the very few particles they consider to be HIV. But if you look at the bottom section, which is prepared from non-infected cells, there are three particles which we have outlined in blue, which look similar to those the authors consider as HIV.
Looking at the sections (a) and (b) it is difficult to decide exactly what criteria the authors to classify particles as HIV. And whatever these criteria may have been, the particles do not have all the morphological characteristics which retroviral particles should have.
One of the first and unambiguous observations one can make about the arrowed particles is their diameter. We have measured all these particles with a micrometer and their average diameter is 136 nM and no particle has a diameter less than 120 nM. Hence these particles are too large to fit the definition of a retroviral particle. Recall that the retroviral diameter is 100-120 nanometres**.
Another matter is that the chemicals used in the cultures depicted in sections (a) and (b), like those we have seen used on previous slides, including the mitogenic agent PHA, were not used in the cultures in the bottom section. That means that the cultures were not treated equally apart from the addition of “HIV”. Hence the bottom section is not a proper control. Given that retroviral particles can appear even spontaneously, or be induced by culture conditions, because the authors did not add these chemicals, including PHA, the possibility cannot be excluded that the appearances in the bottom section may have otherwise turned out identical to sections (a) and (b).
EXTRA
The caption to this EM reads:
FIG. 2. Purified HIV-1 preparations are contaminated by cellular vesicles. Purified vesicles from infected H9 cells (a) and activated PBMC (b)
supernatants (fraction 6 from Fig. 1a and fraction 5 from Fig. 1b, respectively) or from noninfected H9 cells (c) (fraction 6 from Fig. 1e) were treated
for electron microscopic analysis as indicated under Materials and Methods. The cellular vesicles appear to be a heterogeneous population of
both electron-lucent and electron-dense membrane delineated vesicles ranging in size from about 50 to 500 nm. (Original magnification 136,000).
Virions are indicated by arrows.
*Affiliations of the Franco/German collaboration
Centre d’Immunologie de Marseille-Luminy, Case 906, 13288 Marseille, France; and †Robert Koch Institute, Nordufer 20 D13353, Berlin, Germany
** In the revised 2000 taxonomy of retroviruses, under the heading “Morphology”, “Virions are spherical, enveloped and 80–100 nm in diameter”. Hence the diameter of the Franco/German “HIV” particles is even further removed from the defining diameter.
See http://www.virustaxonomyonline.com/virtax/lpext.dll/vtax/agp-0013/rtr03/rtr03-sec1-0001?f=templates&fn=document-frame.htm&2.0#rtr03-sec1-0001

None of the particles in the Franco-German study have knobs or a cone-shaped core or the lateral bodies which are easy to see in the left side image and are also indicated in the right side image. Which means the Franco/German particle cannot be a retrovirus.
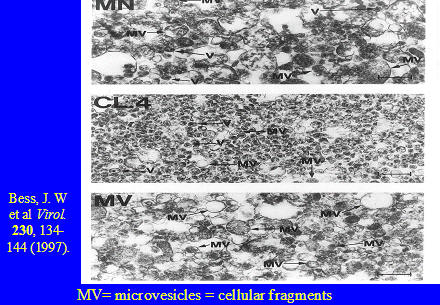
Now we look at the results published by Dr. Julian Bess and his colleagues from America.*
This electron micrograph, also in three sections, represents their efforts to purify HIV.
The top section labeled MN is density gradient material obtained from infected H9 cells. The middle section labeled CL4 is an infected clone of H9 cells called CL4. This culture originated from another culture which was drastically manipulated, including being co-cultured with cells which had been heavily irradiated and were more or less dead. The bottom section labeled MV again represents a density gradient band from a non-infected culture.
Again, as you can see, nothing is purified. In fact it is difficult to discern any differences between the three sections, although CL4 appears to have more particles. In MN and CL4 it is very difficult to find particles with the morphology of retroviruses.
What is labeled MV are microvesicles. There are cellular structures, not retroviral particles and most of the material is made up of microvesicles and other cellular material.
EXTRA
*Affiliations of the American authors
AIDS Vaccine Program, SAIC, National Cancer Institute-Frederick Cancer Research and Development Center, Frederick, Maryland 21702-1201
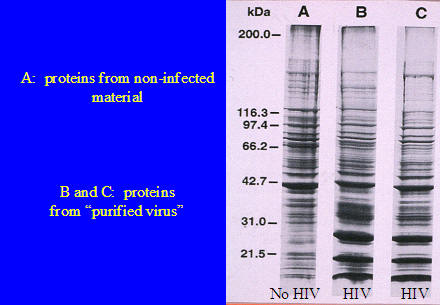
Bess and his colleagues also did an experiment not previously done.
They took the proteins from the three bands, MN, CL4 and MV and transferred them into a gel. That is, they had a gel containing all the proteins in MN, another with all the proteins in CL4 and a third with all the proteins in MV. Then they subjected each gel to an electric field in order to separate the proteins in each gel from one another. This is a standard laboratory technique known as electrophoresis. In this procedure about a 100 volts positive is applied at the bottom of the Lane and because proteins carry a negative charge they begin to move downwards through the gel. The higher molecular weight proteins move slowly and least while the lightest proteins move faster and farthest. After several hours the proteins become separated according to their molecular weights and charge. Then the gels are stained with a protein specific stain which shows the individual proteins as a series of black, horizontal lines. Note that the thickness or darkness of a band is determined by how much protein is in the band. There may be more than one protein in a band. All that is needed is for one or more proteins to have the same or nearly the same molecular weights. The proteins are identified by marker proteins of known molecular weights which are inserted on the left side of the slide. Lane A is the uninfected band, Lane B the infected H9 cells band and Lane C the infected CL4 band.
Our interpretation of this electrophoresis experiment is there is no difference between the three Lanes except that, in some parts, the darkness of the bands varies. (Take care not to confused electrophoretic bands, which are proteins, with density gradient bands, which are parts of a density gradient bearing the same density and hence capable of concentrating material of the same densities. The same word is used for two completely different things).
In other words, in our view the same proteins with the same molecular weights are present in all density gradient banded specimens. Infected and non-infected. The only difference between all these bands is quantitative, not qualitative.